
Freek J van Sluijs, DVM, PhD, DECVS
Gert ter Haar, DVM, PhD, DECVS
Jolle Kirpensteijn, DVM, PhD, DECVS, DACVS
1.1 Introduction
Surgery, literally meaning “handwork” from its Greek origins, is the art of treating a patient for a medical condition by using manual and/or mechanical methods. In doing so, the surgeon attempts to achieve a particular medical outcome. Surgical skills alone are not enough to achieve the intended outcome, and other factors surrounding both the patient and the owner also play an important role. The well-known expression “the operation was a success, but the patient died” may lead one to believe that surgery is purely the act of performing an operation. This, however, is rather short sighted because many more factors (including ethical and economic) are involved in the surgical process before, during and after the operation, which may last weeks to months after the actual procedure itself (Table 1).
Table 1. Factors influencing surgical outcome
Patient-related factors
- Age
- Bodyweight and condition
- Concurrent disease
Surgeon-related factors
- Skills
- Training
- Experience
Practice-related factors
- Surgical theatre
- Personnel (surgery and anaesthesia)
- Hospitalisation facilities
Client-related factors
- Education
- Finances
Age, a patient-related factor, is not, as such, a reason to refrain from surgery, but very young and very old patients present special considerations. Young patients have an increased risk of developing hypothermia and hypoglycaemia, whilst geriatric patients may suffer from a subclinical organ dysfunction, which may affect convalescence. Wound healing may be delayed in patients with severe malnutrition and obese patients have a higher risk of complications during and after surgery. Concurrent diseases such as heart disease or renal failure may increase anaesthetic risk and should be identified and managed before surgery.
Surgeon-related factors are based around the skills of the surgeon, the level of training and experience with the procedure that will be performed.
Practice-related factors include the surgical theatre (overpressure ventilation, electrical safety, management of hygiene), the personnel (presence of specialists or technicians qualified in anaesthesia, adequate surgical assistance) and facilities to hospitalize patients requiring prolonged or intensive care.
Client-related factors are the ability of the client to understand the procedure and instructions for appropriate aftercare, and the willingness or ability to pay for the costs of treatment.
1.2 Clinical decision-making
Clinical decision-making in surgery is primarily focused on the question of whether an operation is the best treatment for the disease concerned. The risks and benefits of an operation must be weighed against the risks and benefits of other therapies. Attention should also be paid to treatments that may support or expand the effect of surgery (physiotherapy, radiation therapy, chemotherapy). Ideally, this assessment is based on the proven efficacy of eligible treatments. In veterinary medicine the cost of treatment is an important factor that must also be taken into consideration. A stepwise approach can help to weigh the costs against the expected results at critical stages in the decision-making process. A diagnostic and therapeutic scenario (or check list) is a detailed description of such an approach. In the scenario all steps of the procedure are described in the sequence in which they should be performed. The scenario may be represented graphically in an algorithm or flow sheet.
Diagnostic and therapeutic scenarios are based on evidence from clinical trials and are developed by scientific professional organizations with input from scientists and practicing veterinarians. When a professional organization has published a scenario, it should be binding for all professionals. Disciplinary judges will use the scenario as a standard for professional conduct to which the action of veterinarians is compared.
Unfortunately, evidence-based scenarios are still rare in veterinary medicine. But in the absence of formal scenarios, veterinary surgeons may develop their own algorithms. An example is given in Figure 1, which shows an evidence-based scenario for the diagnosis and treatment of anal sac carcinoma in the dog.
The algorithm starts after an anal sac adenocarcinoma has been diagnosed by fine needle aspiration biopsy. Additional tests are undertaken (chest X-ray or CT, abdominal ultrasound) to stage the disease. A stepwise approach is followed to ensure that each decision is based on a realistic expectation of the results that can be achieved. These are derived (for this example) from two clinical studies that describe survival in 113 and 130 dogs with anal sac carcinoma.1, 2 In these studies, survival was correlated to the clinical stage with median survival times ranging from 2 (stage 4) to 40 months (stage 1). The algorithm illustrates that in advanced stages more treatment modalities are necessary to obtain shorter survival times. The expected survival may help to balance the costs of treatment against the outcome. The algorithm also depicts the sequence in which decisions are made.
Stage 1 is the simplest situation: a small tumour without regional or distant metastasis. Treatment consists of one modality (resection of the tumour) and is expected to produce good long-term results. In stage 2 the tumour is large (> 10 cm2) but there are no regional or distant metastases. If the tumour cannot be removed it may be treated by radiation to reduce the size until it has become resectable. Life expectancy after treatment is less than in stage one, but still acceptable. Stage 3 is characterized by regional metastases from a small or large tumour. If the metastases are resectable, they will be removed; the same applies to the primary tumour. Radiation therapy will be used preoperatively or intra-operatively if the metastases or the tumour are too large for removal or postoperatively. When their size has been reduced sufficiently, the tumour and the metastases are resected. The costs in this scenario increase with the number of treatments, whilst the life expectancy decreases to half of that in stage 2. In stage 4, the disease has spread to distant sites. Life expectancy is short, and treatment is mainly palliative.
The advantage of the algorithm is that it provides insight into the costs and benefits of treatment in various stages of the disease. This helps the veterinarian and the owner to balance these aspects. The algorithm’s limitation is that it displays median survival of a group of patients – individual patients may have a longer or shorter survival. One should be aware of this limitation when using algorithms.

Fig. 1. Diagnostic and therapeutic algorithm for anal sac carcinoma in the dog
An important aspect in the anal sac adenocarcinoma example is the assessment whether the tumour (or the lymph node) is resectable. In general, tumours are resectable if they can be removed with safe margins without causing damage to nearby vital structures (Figure 1). In the anal sac adenocarcinoma example, nearby vital structures are the external anal sphincter and the pudendal nerve. The close proximity of the sphincter to the anal sac excludes resection with wide margins; however, the tumour is resectable if it can be removed with narrow margins without causing severe damage to the sphincter. Vital structures near enlarged lymph nodes may include the rectum and colon, the urinary bladder, prostate and ureters, the aorta and the external and internal iliac arteries.
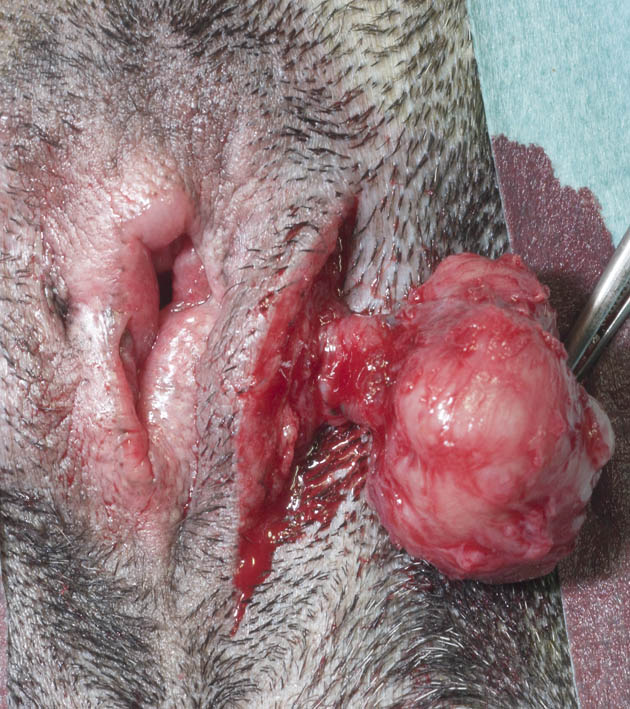
Figure 2. Removal of a tumor in the anal sac with safe margins
Before asking whether or not the tumor is resectable, the surgeon must assess whether the patient is operable. The American Society of Anesthesiologists has developed a qualification system for the physical status in human patients that is also used in veterinary medicine. Patients are divided into 5 classes:
Table 2. ASA classification of anaesthetic risk[*]
I A normal healthy patient
II A patient with mild systemic disease
III A patient with severe systemic disease
IV A patient with severe systemic disease that is a constant threat to life
V A moribund patient that is not expected to survive without the operation
Patients in category I-IV are usually operable, provided that sufficient knowledge, experience and equipment are available to deal with complications. In group V, there is little choice but this category may be decreased to a lower grade by providing adequate supportive therapy. An example of this is hyperkalemia in a cat with urethral obstruction. If a catheter can be inserted, fluid therapy may move the patient from category V to category IV or III and reduce the risk of cardiac arrest and other problems during surgery and anesthesia.
1.3 Purpose of surgery in animals
- Prophylactic surgery: for instance, to prevent neoplastic disease, e.g. ovariectomy (OVE) for mammary gland tumours, excision of actinic dermatitis from the auricle or nasal plane for squamous cell carcinoma (SCC), and castration for cryptorchid testicles.
- Therapeutic surgery: in the treatment of e.g. wounds, lesions, inflammation, anatomical changes, tumours
- Palliative surgery: to improve but not completely cure the disease that the animal is afflicted with (e.g. debulking surgery)
- Diagnostic: e.g. biopsy, operations which reveal a disease (like endoscopic exploratory surgery or an exploratory laparotomy)
- Surgery to increase the animal’s utility for certain purposes: e.g. castration, dehorning
- Experimental surgery for biomedical research
1.4 Methods for cutting or destroying tissue
- Cutting with sharp instruments
- Cutting or destroying tissue using high frequency currents: electrosurgery (‘electric knife’) and radiosurgery
- Destroying tissue by freezing (and thawing): cryosurgery or cryonecrosis
- Destroying tissue by the direct application of hot metal or by chemicals: cauterisation
- Localised thermal effect using Light Amplification by Stimulated Emission of Radiation (‘laser’ surgery)
- Ultrasonic ablation: e.g. Ultrasonic Surgical Aspirator (CUSA)
- Controlled ablation (coblation)
- Cutting with radioactive devices (focused radiation therapy including gamma knife)
The latter three devices are rarely available for general veterinary practice due to costs but can be used in specialized (university or private practice) clinics or research facilities. Methods 1-5 will be discussed in length in the upcoming chapters.
1.5 Methods to control intra-operative haemorrhage
- Pressure
- Mechanical devices (sutures, clips and ties)
- Coagulation devices
- Laser devices
- Sealing devices (e.g., the LigaSure, ENSEAL)
- Drug carrying devices, e.g. gel foam or biomatrix
1.6 Nomenclature
The best way to name operations is to use a combination of anatomical terms and Greek suffixes, thereby describing the location and type of operation. Exceptions are operations named after the inventor or surgeon. Examples:
–tomy (cutting):
- thoracotomy: opening the thorax
- gastrotomy: incising the stomach
–stomy (making an opening):
- colostomy: making an artificial opening of the colon on the surface of the abdomen
- gastroduodenostomy: creating an anastomosis between stomach and duodenum
–ectomy (excision):
- splenectomy: removing the spleen
- ovariohysterectomy: removing the uterus and the ovaries
- ovariectomy: removing the ovaries
- orchidectomy: removing the testicles (castration)
–plasty (shaping, forming):
- episioplasty: reconstruction of the vulva
–centesis (perforating or draining):
- paracentesis: penetrating a body cavity for the aspiration of liquid
- cystocentesis: penetrating a urinary bladder for the aspiration of liquid
–pexy (attaching):
- gastropexy: attaching the stomach to the abdominal wall to prevent torsion (volvulus)
- colopexy: attaching the colon to the abdominal wall to prevent a rectal prolapse
–rrhaphy (suturing or closing):
- inguinal herniorrhaphy: suturing an inguinal hernia
- diaphragmatic herniorrhaphy: suturing a diaphragmatic hernia
- perineal herniorrhaphy: suturing a perineal hernia
- temporary tarsorrhaphy: temporarily suturing upper to lower eyelid
It should be noted that the field of surgery extends beyond the surgical procedure alone. Many other therapeutic modalities are used too, such as wound dressings, pharmacotherapy, radiation- and physiotherapy, etc.
1.7 References
1. Williams LE, Gliatto JM, Dodge RK. (2003) Carcinoma of the apocrine glands of the anal sac in dogs: 113 cases (1985-1995). Journal of the American Veterinary Medical Association, 223(6):825–31
2. Polton GA, Brearley MJ. (2007) Clinical stage, therapy, and prognosis in canine anal sac gland carcinoma. Journal of Veterinary Internal Medicine, 21(2):274-80
3. https://www.acvs.org/small-animal/anal-sac-tumors and https://vsso.org/anal-sac-tumors (not peer-reviewed documents)
[*] The original qualification has a sixth category that is not applicable to veterinary medicine: a brain-dead patient whose organs are removed for donor purposes.
Bio Prof Freek van Sluijs
1993 Diplomate, European College of Veterinary Surgeons
1987 PhD: Gastric Dilation-Volvulus in the Dog
1991 – 1998 ECVS – Founding member and President
1993 – 1999 EBVS – Founding member and President
1990 – 2012 Professor of Companion Animal Surgery, Department of Clinical Sciences of Companion Animals, Faculty of Veterinary Medicine, Utrecht University
2012 – 2015 Deputy Professor of Companion Animal Surgery, Klinik für Kleintierchirurgie, Vetsuisse Faculty, University of Zürich
